

 |
|||
 |

|

|
 |
The interest in Bombyx mori silk emerges from its biocompatibility and its structural superiority to synthetic polymers. Silk fibers have been used for millenia because they are strong, and the microscopic origin of that strength is well understood. Recently, the ability to chemically reconstitute cocoons back into solution and transform that solution into new forms has created a new class of silk based materials. The reconstitution of silk is outlined below, where you: 1) begin with silk cocoons, 2) isolate the fibroin protein, and 3) resolubilize the protein by lithium denaturation. With these new materials comes a need to associate their structure with bulk mechanical properties. My research involves turning reconstituted silk protein solutions into gels by changes in pH.
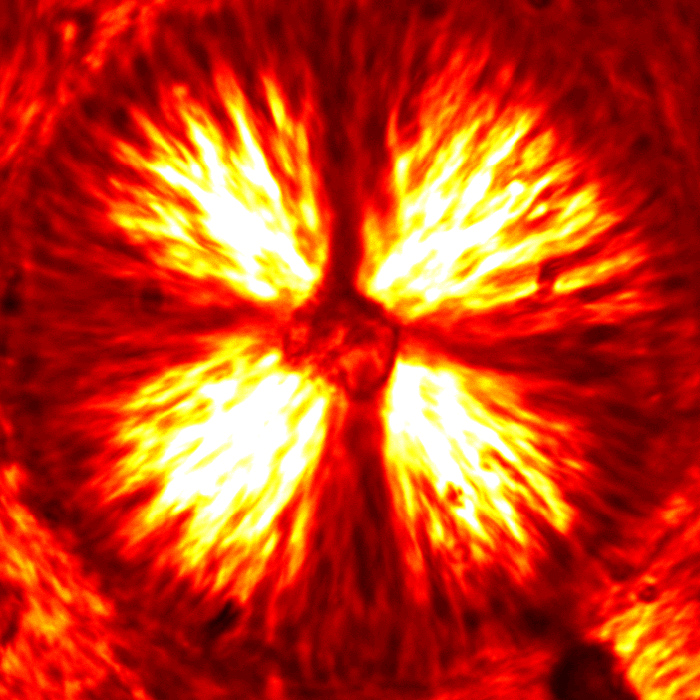
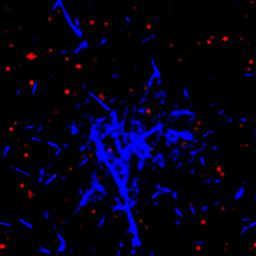
Depending on the concentration of acid added to the system, silk protein will assemble into aggregates or network spanning structures. The video below shows the kinetics of pH induced gel formation. A small volume of high concentration acid is added to a silk solution out of the frame (to the bottom right), and the acid will diffuse into the frame. When this happens, the protein phase separates and eventually forms a spanning network. Your eye might pick out a characteristic spherical size for the aggregates before they merge with others to create a percolated network. It turns out that this aggregate characteristic size is strongly dependent on the acid concentration.
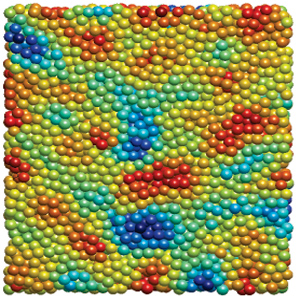
These aggregates are composed of silk fibroin protein. And while the aggregates interact to form a network, the fundamental interaction in the system is between two proteins. This is not an easily accessible length scale, but we have chosen to use small angle neutron scattering (at the National Institute for Standards and Technology in Gaithersburg, Maryland) to probe the structure of these proteins in various stages of aggregation; probing structure is an indirect probe of the interaction between protein. A result is shown below where the arrow signifies increasing the acid concentration.
By combining microscopy, light scattering, and neutron scattering, we can span a huge range of length scales and quantify the aggregation of reconstituted silk protein at both the aggregate and individual protein scale.

For more information, please contact Pasha Tabatabai or visit Pasha's website.